
+1 804 381 0905
+44 1233 638 383
+86 021 585 854 63
user/addons/gmaps/mod.gmaps.php, line 63 show details
ee/EllisLab/ExpressionEngine/Boot/boot.common.php, line 539 show details

+1 804 381 0905
+44 1233 638 383
+86 021 585 854 63
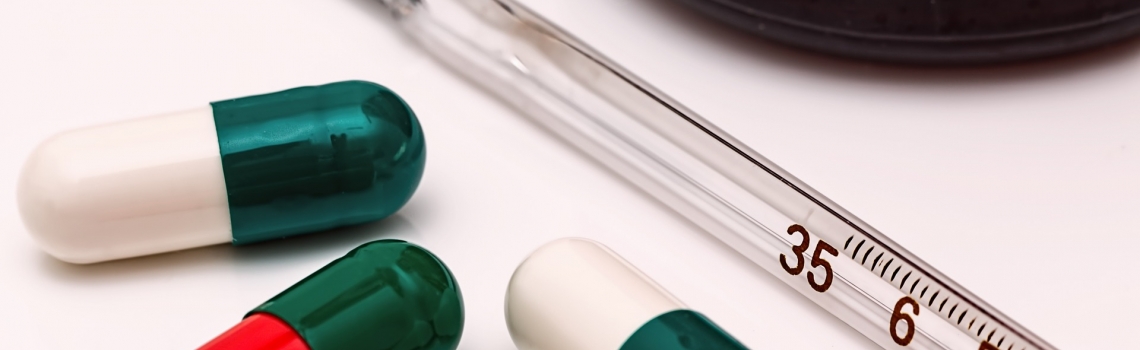
Introduction
Antibiotics have transformed the treatment of disease since the discovery of penicillin in 1928.
In the 20th century, antibiotics enabled the treatment of what had previously been common and fatal infectious diseases such as tuberculosis and, since then, they have played a significant role in modern healthcare. However, the rise of antibiotic resistance has sparked fears that treating bacterial infections may soon no longer be possible. According to the lowest estimates, antimicrobial resistance causes around 700,000 deaths every year and that figure could increase to 10 million per year by 2050.
Older medicines are becoming increasingly ineffective against microbes and there are not enough new medicines in the pipeline to combat resistant organisms. Enhancing how we use antibiotics in order to handle the emerging resistance is therefore a key strategy for easing the pressure to develop new antimicrobials and for reducing the risks that resistance poses to global health.
Bedside diagnostics
Coming under the umbrella of ‘antimicrobial stewardship’ are strategies to improve antibiotic use, with the focus being on selecting the most suitable antibiotic, dosage and duration of treatment for the specific infection. However, a lack of rapid diagnostics for guiding clinical decisions about the correct antibiotic to use, means the ability to achieve this is somewhat limited.
The UK Prime Minister commissioned the Independent Review on Antimicrobial Resistance in order to advise which actions should be taken to combat global antimicrobial resistance. In the review, rapid diagnostic tests are described as key to enacting behaviour change in the prescription of antibiotics.
The review panel calls for these “game-changing” diagnostics to be adopted on a widespread basis between now and 2020 in order to realise a “right patient, right antibiotic, right time” approach to antibiotic prescribing.
“If the world is serious about tackling the threat of drug-resistant infections, we need to fully embrace the step-change in technology that rapid point-of-care diagnostics represent,” the review concluded. “Only by doing this can we fundamentally and sustainably reduce our misuse and overuse of antibiotics.”
Automation with the Ther-Mix
While the ETGA technique is significantly faster than conventional blood culture, it can be made even faster through automation.
Researchers at Momentum Biosciences use the Ther-Mix from Vitl Life Science Solutions at the heart of the ETGA method to streamline their techniques. The Ther-Mix enables the team to semi-automate the workflow, which delivers results even more quickly, frees up bench space, reduces hands-on labor, and improves standardisation of the techniques.
With the help of the Ther-Mix, a single unit can perform several mixing and incubation steps, with the capability for as many as 100 customized mixing programs to be plugged in from beginning to end, thereby enabling operators to walk away and leave the Ther-Mix to do the work.
References
Crow MA, Bennett HV, Grover M, et al. Evaluation of a new method for the rapid exclusion of infection in suspected bacteraem. Presented at: 24th European Congress of Clinical Microbiology and Infectious Diseases; 10-13 May 2014. Barcelona, Spain.
Dryden M. Near-patient testing, infection biomarkers, and rapid diagnostics. In: Laundy et al. (eds.) Antimicrobial Stewardship. Oxford University Press, 2016.
Kopnitsky M (2013). A new twist on the detection of disease-causing microbes. Available at: http://www.mlo-online.com/a-new-twist-on-the-detection-of-disease-causing-microbes.php
O’Neill J, chair (2014). Antimicrobial resistance: Tackling a crisis for the health and wealth of nations. Available at: http://amr-review.org/Publications
O’Neill J, chair (2015). Rapid diagnostics: Stopping unnecessary use of antibiotics. Available at: http://amr-review.org/Publications
Zweitzig DR, Sodowich BI, Riccardello NM, et al. Feasibility of a Novel Approach for Rapid Detection of Simulated Bloodstream Infections via Enzymatic Template Generation and Amplification (ETGA)eMediated Measurement of Microbial DNA Polymerase Activity. Journal of Molecular Diagnostics 2013; 15: 319-330.